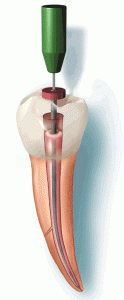
The first visit to the dentist, the dentist will gives you local anaesthesia and remove all the pulp tissue. He will then put a temporary filling material to fill up the empty spaces. This process is called Access Opening. During the second visit, the temporary filling material is removed and root canal material like gutta percha is placed. A temporary filling material is placed again. This process is called Obturation. During your last visit, the temoporary filling material is removed and replaced by permanent filling material like composite/amalgam and a crown preparation is done.
Definition
“ The three dimensional filling of the entire root canal system as close to the cementodentinal junction as possible.â€
– American Association of Endodontist
A large variety of root canal filling materials have been advocated through various decades. The gamut of materials run from plaster of paris, bamboo, gold, iridioplatinum to most, silver cones to most popularly as gutta-percha.
In 1847, Hill developed a preparation. It consisted principally of bleached gutta purcha and carbonate of lime quartz. It was popularly called Hill’s stopping. This was a very crude form when first introduced, and it was patented in 1848.
The function of root canal filling is to obturate the canal and eliminate all portals of entry between periodontium and the root canal . It should provide hermetic seal.
Objectives:
3D obturation serves following objectives
1) It provides inert substitute for pulp which was previously occupying the space.
2) It prevents percolation and micro leakage of periapical exudate into Rc space
3) Prevents re-infection
4) Creates biologic environment for healing of periapical tissue
When to obturate the canal :
1 ) Tooth should be asymptomatic
2 ) Canal is dry
3) No Sinus tract
4) No foul odour
5) Temporary filling is intact
6) Culture test is negative
7) No tenderness of tooth
8) No pain on palpation of oral mucosa
 Ideal requirements of Root Canal material
 1) The material should be easily introduced in the canal
2) It should seal laterally as well as apically
3) It should not shrink after insertion
4) It should be impervious to moisture
5) It should be bactericidal
6) It should be radiopaque
7) It should not stain the tooth structure
8) It should not irritate tissue
9) It should be sterile or easily sterilized before insertion.
10) It should be easily removed from Root Canal if necessary
Root canal Materials are classified into:
1.Orthograde
2.Retrograde
Orthograde filling materials are classified as:
1. Solid : Semi rigid : Silver cones, Stainless steel
Rigid : Vitallium, Cr-Co implant cone
2. Semisolid
Gutta-Percha
3. Pastes
a)Â ZnO
b) Resins
4. Combination of solid and semisolid
Retrograde materials are
Amalgam,GIC,super EBA,IRM,Cavit,composites
MTA, DBA, Calcium phosphate cement.
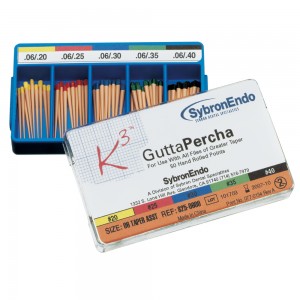
Gutta-percha:
Is the dried juice of Taban tree (Isonandra percha) It was introduced by Bowman in 1867. It occurs naturally as 1,4 –poly isoprene and is harder, more brittle and less elastic than natural rubber and hence plasticizers are added to improve the properties.
It exists in 2 crystalline forms :
1) a Phase (soft)
Interchangeable
2) b  Phase (hard)
a Form is the material that comes from natural tree
b form is the processed form that is used for obturation
(conventional GP points)
– b form is commercially available form
Newer products have adopted a crystalline structure.
* When there is increase in temp – b  – phase
a – phase at 460C [37-49]
g   – phase at 54-600C (Amorphous phase)
Composition :
Gutta-percha 20% [Matrix]
Zinc oxide 60- 75% – (Filler) antimicrobial
Resins, waxes 5-10% – (Plasticizer)
Metal sulfates 11% provides radiopacity
Coloring agents
G.P is manufactured in
Standardized sizes(coordinate with ISO size)
Non standardized sizes
(more tapered, used as auxiliary cones )
Designated as extra fine, fine –fine
Medium fine, fine
Fine medium, medium
Medium large, large
PROPERTIES OF GP
GP has property of excellent adaptation to the walls of canal
GP is least reactive and so biocompatible material
No dimensional change after condensation in the canal
GP is well tolerated in tissue and does not encourage bacterial growth
GP is radiopaque
GP can be dissolved by solvents such as chloroform, xylol, eucalyptol
GP does not discolor the tooth structure
G.P dissolves in chloroform and eucalyptus oil
Disinfection of G.P by immersing it in 5.25% Naocl for 1min (kills vegetative organisms and spore forms
Medicated G.P has iodoform Component
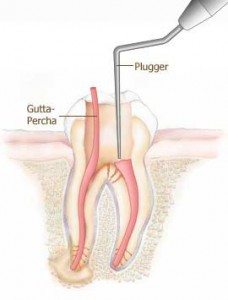
Rejuvenation:
G.P has limited shelf life.
It becomes brittle with age, so rejuvenation is done by momentary immersion in hot tap water (550C) followed by instant cooling by cold tap water.
Silver Cones:
Composition:
Silver 99.4%
Nickel 0.4%
Copper 0.2%
Advantages:
More accurately standardised and calibrated than G.P
can be easily placed in small curved canals.
Does not shrink and easily sterilized.
Disadvantages:
Difficulty in retrieving the Ag cone
Poor lateral seal
Corrosion products are toxic.
Properties:
It does not shrink.
It is impervious to moisture.
It does not encourage bacterial growth but may even inhibit it.
It is not irritating to periapical tissue except when grossly extruded through the apical foramen.
It is radioopaque.
It is sterilized quickly and easily over an open flame.